
Never Miss a Beat,
That’s Bioflux
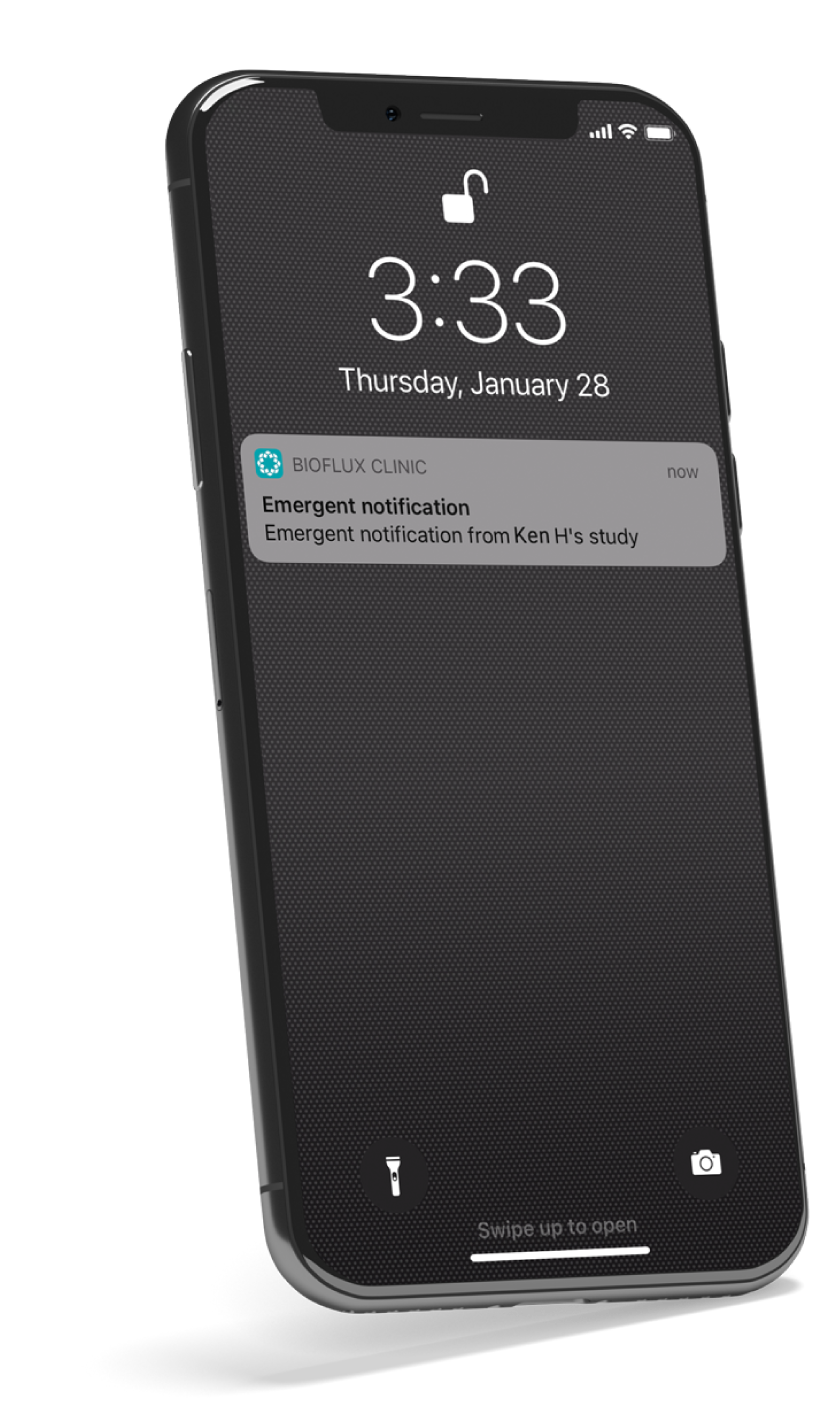
Cardiac issues can be challenging to diagnose when symptoms are intermittent or absent. Identifying abnormal heart rhythms as they happen can lead to early intervention, potentially saving a patient’s life.
Bioflux addresses this problem by capturing and analyzing heart health data on the device. Alerts are available to physicians when Bioflux detects abnormal heart rhythms or when the patient pushes the event button, signaling a symptomatic event.

Faster Diagnosis
Our application, cloud, and wireless technologies connect and share data seamlessly for faster data analysis, leading to faster diagnosis.
Streamlined Workflow & Increased Revenue
Seamless integration with clinic workflows for faster care delivery and insurance reimbursable for practice growth.
Enhances Data Quality and Patient Safety
Continuously collects, analyzes & automatically uploads up to 3 channels of ECG, enabling higher quality and more accurate diagnoses.
Optimized Patient Care
Enhancing the quality of care by combining monitoring, faster diagnosis, and direct-to-home delivery.
Faster Diagnosis
Our application, cloud, and wireless technologies connect and share data seamlessly for faster data analysis, leading to faster diagnosis.
Streamlined Workflow & Increased Revenue
Seamless integration with clinic workflows for faster care delivery and insurance reimbursable for practice growth.
Enhances Data Quality and Patient Safety
Continuously collects, analyzes & automatically uploads up to 3 channels of ECG, enabling higher quality and more accurate diagnoses.
Optimized Patient Care
Enhancing the quality of care by combining monitoring, faster diagnosis, and direct-to-home delivery.

Interested in Learning More?
1-800-590-4155
What Sets Us Apart
Comparison Features |
Cardiac Outpatient Monitoring | |
|---|---|---|
| Wearable | Daily Activity | Daily Activity |
| ECG Channels | 3 Channels | 2 Channels |
| Report Turnaround Time | 3 Day Average | 1 Week |
| Remote ECG Access |  |
 |
| Rechargeable |  |
Not Standard |
| Physician Enabled Billing |  |
 |
| Practice Online Portal/App |  |
 |
| Low-Cost Ownership |  |
 |
Swipe to Compare other Methods

Superior Solution
With over 50 billion heartbeats recorded and trusted by over 2,500 health care professionals, we pride ourselves on being an industry leader and innovator in remote cardiac care. Whether your patients’ cardiac issues are symptomatic or asymptomatic, Bioflux is a necessary tool for your clinic. Physicians have insight into patient studies with access to ECG data remotely through the Biocare diagnostic portal, where they can initiate and change patient studies, request a real-time ECG, and receive results. Millions of ECG data points are seamlessly and securely transferred to the Biocare diagnostic portal and app, where you can view patient data at any time. Our mission is to improve care while giving you peace of mind.
Data Uploaded 24/7
Assurance that significant events will be captured and reviewed quickly
3 Channels
Built-in redundancy helps reassure patients that they are getting the best study possible
Small and Lightweight
Easy to use, comfortable and discreet
Need an efficient solution for your practice?
Need an efficient solution for your practice?
The Bioflux Includes
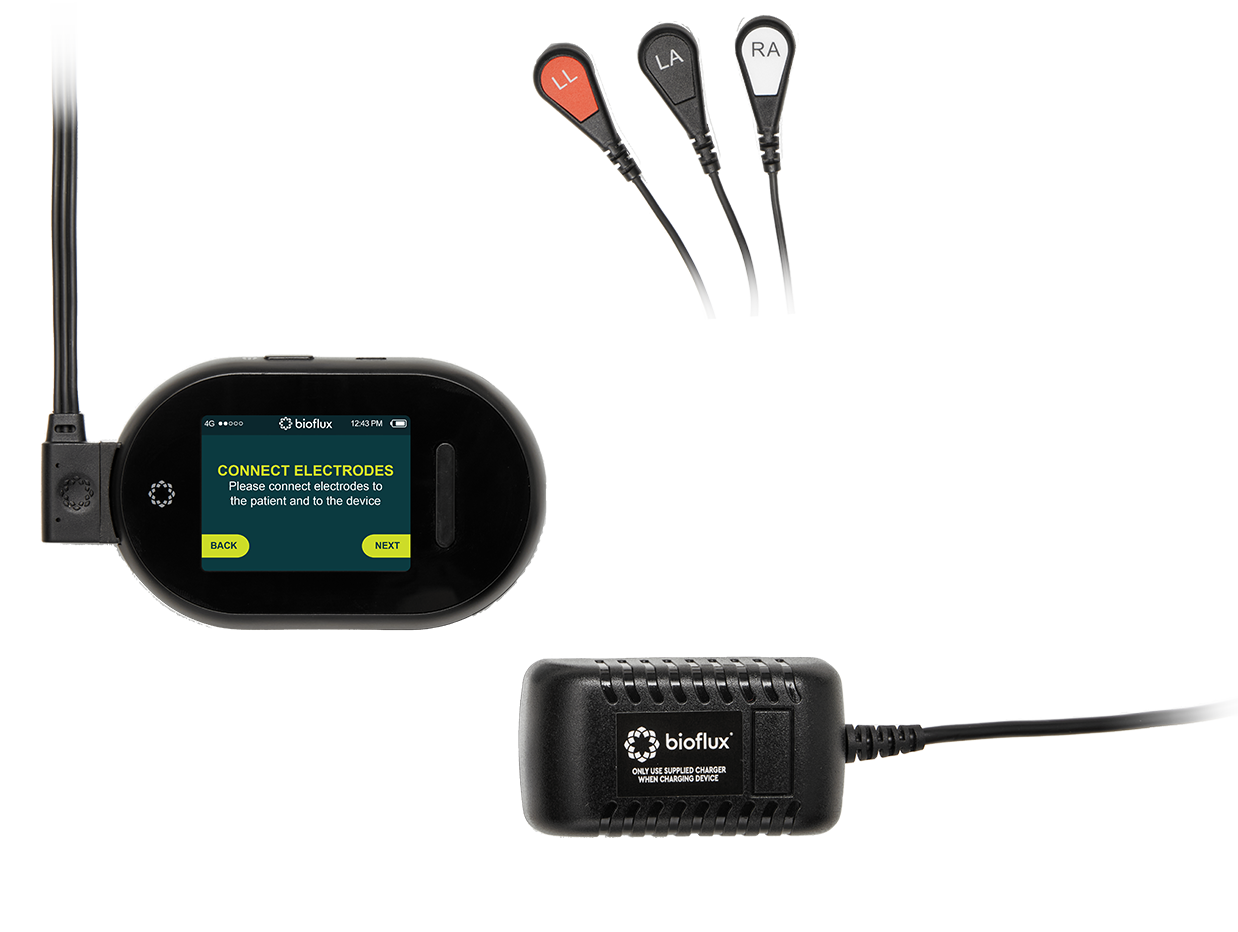
Getting Started
Bioflux is a superior approach to active heart monitoring.
Prescribe Bioflux
Once prescribed, Bioflux is attached to the patient’s chest for heart monitoring. This can be done by the physician’s office, or through Biodirect, where Biotricity sends Bioflux directly to the patient for self-application.


Identify Abnormalities
While users wear the device, Bioflux will identify and flag tachycardia, bradycardia, pause, and atrial fibrillation for review. When users feel symptoms such as palpitations, light-headedness, or unexplained rapid heartbeat, they can press the Event button to flag their symptoms for review as well. Physicians have access to these events if and when they want it.


Review Results
When the study is complete, the patient returns Bioflux to the physician’s office, and the results are already available for review by the doctor.
Biodirect Program
For added convenience, Biotricity can ship Bioflux directly to the patient with instructions on applying and using the monitor.
Hear from Clinicians

“In my transition from a hospital-based practice to a hybrid-practice, Bioflux facilitated remote monitoring of my patients with a wide variety of cardiovascular rhythm complaints leading to definitive diagnoses and targeted management”

“Bioflux is a great product giving 3 channels to detect arrhthmias more accurately. The online portal is easy to navigate for review and interpretations. Reports are ready in 48-72 hours, allowing me to give patients results quickly and efficiently.”

“One of the key benefits with Bioflux monitors is the ability to view the patient “live” ECG at hookup and at different points throughout the study. We use other Holter monitors that don’t allow the technician to see the quality of hookup which could cause the entire study to be affected. Adding Biotricity to our toolbox was a positive move. “







